Answer
378.3k+ views
Hint: The zinc-carbon dry cell consists of a zinc anode and graphite cathode. The electrolyte is in the form of a low moisture paste made up of ammonium chloride and zinc chloride.
Complete Solution :
-First of all let us see what a dry cell is.
- A dry cell is basically a type of an electrochemical cell or a battery which is mostly used for the house equipment and portable electronic devices. The electrolytes present in it are in the form of a paste to form low moisture immobilized electrolytes, which restricts it from flowing and the moisture is low but enough to allow the current to flow. This also makes it easy to transport.
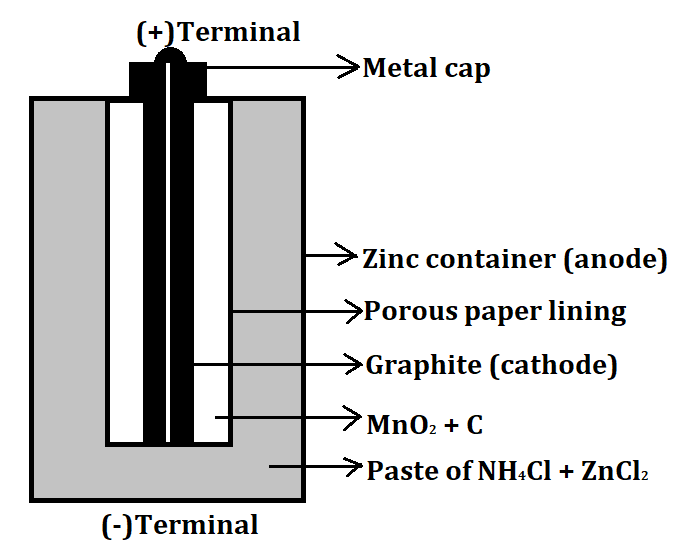
- An example of a dry cell is a Zinc-Carbon cell. It is a type of primary cell and also known as a Leclanche cell. It can produce a maximum of 1.5V of voltage. It consists of a Zinc container which acts as the anode (negative electrode) and a carbon graphite rod which acts as a cathode (positive electrode). The remaining space between both the electrodes is filled with manganese dioxide ($Mn{O_2}$) and a low moisture electrolyte made up of ammonium chloride ($N{H_4}Cl$) and zinc chloride paste ($ZnC{l_2}$).
The overall reaction involved in this cell is given below:
$Zn + 2Mn{O_2} + 2N{H_4}Cl \to M{n_2}{O_3} + Zn{\left( {N{H_3}} \right)_2}C{l_2} + {H_2}O$
The labelled diagram for this dry cell is shown below:
Note: A dry cell is basically of 2 types: primary and secondary. A primary dry cell is the one in which the electrochemical reactions consume all the chemical reagents, thus fail to produce electricity and so are neither reusable nor rechargeable. For example: zinc-carbon cell, lithium cell, mercury cell, silver oxide cell, etc.
- While the secondary dry cell has the ability to regenerate the chemical reactions and hence can be recharged with the help of battery chargers. For example: Nickel metal hydride cell, nickel-cadmium cell, lithium ion cell, etc.
Complete Solution :
-First of all let us see what a dry cell is.
- A dry cell is basically a type of an electrochemical cell or a battery which is mostly used for the house equipment and portable electronic devices. The electrolytes present in it are in the form of a paste to form low moisture immobilized electrolytes, which restricts it from flowing and the moisture is low but enough to allow the current to flow. This also makes it easy to transport.
- An example of a dry cell is a Zinc-Carbon cell. It is a type of primary cell and also known as a Leclanche cell. It can produce a maximum of 1.5V of voltage. It consists of a Zinc container which acts as the anode (negative electrode) and a carbon graphite rod which acts as a cathode (positive electrode). The remaining space between both the electrodes is filled with manganese dioxide ($Mn{O_2}$) and a low moisture electrolyte made up of ammonium chloride ($N{H_4}Cl$) and zinc chloride paste ($ZnC{l_2}$).
The overall reaction involved in this cell is given below:
$Zn + 2Mn{O_2} + 2N{H_4}Cl \to M{n_2}{O_3} + Zn{\left( {N{H_3}} \right)_2}C{l_2} + {H_2}O$
The labelled diagram for this dry cell is shown below:

Note: A dry cell is basically of 2 types: primary and secondary. A primary dry cell is the one in which the electrochemical reactions consume all the chemical reagents, thus fail to produce electricity and so are neither reusable nor rechargeable. For example: zinc-carbon cell, lithium cell, mercury cell, silver oxide cell, etc.
- While the secondary dry cell has the ability to regenerate the chemical reactions and hence can be recharged with the help of battery chargers. For example: Nickel metal hydride cell, nickel-cadmium cell, lithium ion cell, etc.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

What is the stopping potential when the metal with class 12 physics JEE_Main

The momentum of a photon is 2 times 10 16gm cmsec Its class 12 physics JEE_Main

How do you arrange NH4 + BF3 H2O C2H2 in increasing class 11 chemistry CBSE

Is H mCT and q mCT the same thing If so which is more class 11 chemistry CBSE

Trending doubts
Difference between Prokaryotic cell and Eukaryotic class 11 biology CBSE

Difference Between Plant Cell and Animal Cell

Fill the blanks with the suitable prepositions 1 The class 9 english CBSE

Change the following sentences into negative and interrogative class 10 english CBSE

Give 10 examples for herbs , shrubs , climbers , creepers

What organs are located on the left side of your body class 11 biology CBSE

Write an application to the principal requesting five class 10 english CBSE

What is the type of food and mode of feeding of the class 11 biology CBSE

Name 10 Living and Non living things class 9 biology CBSE



